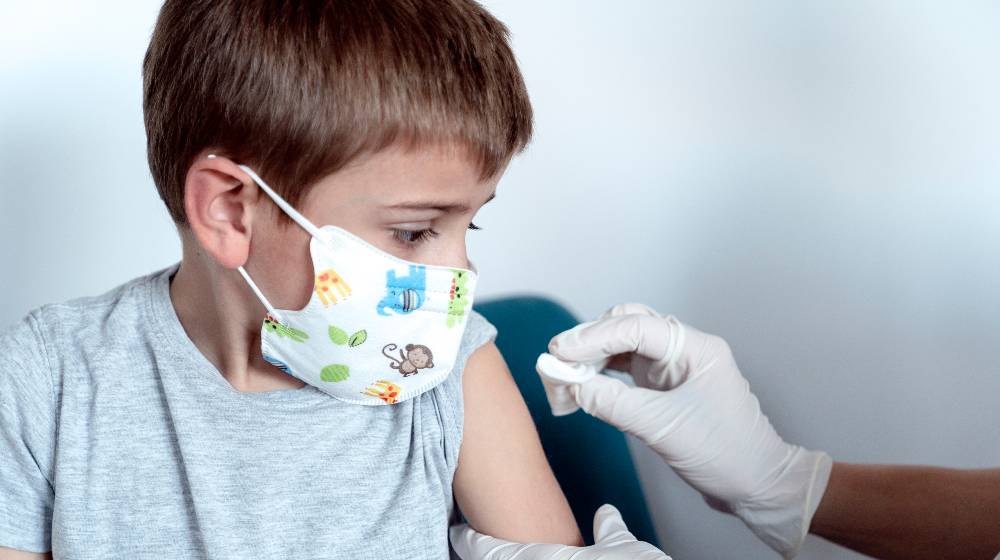
Pfizer and BioNTech filed an emergency use authorization request to allow them to start vaccinating for COVID-19. The joint developers of a coronavirus vaccine submitted the request to the US Food and Drug Administration in order to inoculate Americans aged 5 to 11 years.
RELATED: Are You Ready For COVID-19 Vaccine For Children?
Pfizer Wants Emergency Use Authorization To Vaccinate Kids from COVID-19
 In a tweet sent yesterday, Pfizer announced their application for emergency use authorization. “With new cases in children in the U.S. continuing to be at a high level, this submission is an important step in our ongoing effort against #COVID19. We’re committed to working with the FDA with the ultimate goal of helping protect children against this serious public health threat,” the drugmaker posted. As a result of Pfizer’s request, an FDA advisory committee will meet on October 26 to discuss authorization. If approved, Pfizer and BioNTech’s coronavirus vaccine would become the first that’s specifically okayed for use by kids under 12 years. Last month, both companies said that their studies showed that the vaccine proved safe and effective for children aged five to eleven. Based on data that involved trials in more than 2,000 children, the vaccine provided a robust antibody response.
In a tweet sent yesterday, Pfizer announced their application for emergency use authorization. “With new cases in children in the U.S. continuing to be at a high level, this submission is an important step in our ongoing effort against #COVID19. We’re committed to working with the FDA with the ultimate goal of helping protect children against this serious public health threat,” the drugmaker posted. As a result of Pfizer’s request, an FDA advisory committee will meet on October 26 to discuss authorization. If approved, Pfizer and BioNTech’s coronavirus vaccine would become the first that’s specifically okayed for use by kids under 12 years. Last month, both companies said that their studies showed that the vaccine proved safe and effective for children aged five to eleven. Based on data that involved trials in more than 2,000 children, the vaccine provided a robust antibody response.
Smaller Doses For Kids
The child test subjects were given two smaller doses compared to regular doses. Pfizer and BioNTech said that the vaccine produced the necessary antibodies. It also produces side effects consistent with studies made in people aged 16 to 25. Pfizer’s vaccine already received full approval status last August. Previously, it applied for emergency use authorization last November. Until now, the Pfizer and BioNTech vaccine remain the only ones given full approval by the FDA for people ages 16 and older. For 12 to 15-year-olds, the FDA gave the same vaccine emergency use authorization only.
Delta Variant Affecting Children
The request for emergency use authorization for kids comes at a time when the highly contagious Delta variant continues to run wild across the US. Whereas previous strains seemingly did not affect children, the delta variant is now leading to an increased number of cases for children. Pfizer said that the ineligibility of vaccines to younger children may have payed apart. Since last year when the pandemic started, around 6 million American kids tested positive for the virus. This is according to the American Academy of Pediatrics. Pfizer and BioNTech hope that a ruling comes out by November.
Not All Parents Okay With Vaccinating Kids
In a Kaiser Family Foundation survey dated September 30, only a third of American parents would vaccinate their 5 to 11 children right away once the FDA issues an emergency use authorization. Another third would wait and see before committing their kids. 24% of parents remain adamant that they won’t vaccinate their younger children. Kids who contract COVID-19 get significantly less sick from the virus compared to adults. Only on very rare occasions are there severe or fatal cases. Watch the ABC News video reporting that Pfizer applies for authorization of vaccines for kids 5 to 11 years old:
Do you support an emergency use authorization to vaccinate 5 to 11-year-olds kids from coronavirus?
Will you get your children vaccinated immediately once Pfizer gets an emergency use authorization? Or, will you wait it out until we get a clearer understanding?
Let us know what you think. Share your comments below.